[E8] Moderna: A vision for medicine using synthetic biology
Transformative vaccines and medicines from mRNA as the "software of life"
Dear Reader,
This profile in Product | Strategy | Innovation provides insights into the science, leaders and company who designed a vaccine in 48 hours and launched it commercially 10 months later using synthetic messenger RNA (mRNA) to help the world battle a pandemic. But beyond vaccines for infectious diseases, the company is pursuing therapies for cancer, cardiovascular, autoimmune and rare diseases on its product roadmap. Scaled production to deliver 800 million to 1 billion doses of the Moderna COVID-19 Vaccine by the end of 2021 and 3 billion by the end of 2022 plus the safety data generated on such a large vaccinated population could accelerate the pipeline for future therapies on this novel platform.
Moderna was founded in 2010 by Harvard, MIT and Flagship Ventures (now Flagship Pioneering) scientists to commercialize synthetic biology using modified mRNA technology to develop vaccines and novel drugs. Derrick Rossi was a postdoctoral fellow in stem cell biology at Stanford University in 2005 when he read about the synthetic mRNA research at the University of Pennsylvania by Drew Weissman and Katalin Kariko. Rossi pursued stem cell research in 2007 as a new assistant professor at Harvard Medical School running his own lab to build on the synthetic mRNA technology from the University of Pennsylvania. Rossi recruited a postdoctoral fellow Kenneth Chien to pursue reprogramming adult stem cells with synthetic mRNA to act like embryonic stem cells.
After more than a year of work, Chien showed modified adult stem cells behaving like embryonic stem cells under a microscope to Rossi who then informed Timothy Springer, another professor at Harvard Medical School and biotechnology entrepreneur. Springer recognized the commercial potential of the stem cell research and contacted MIT biomedical engineering professor Robert Langer who is also a prolific inventor and expert on drug-delivery technology. Rossi, Springer and Langer met in May 2010 at Langer’s MIT lab in Cambridge, Massachusetts. The meeting led to discussions around developing new drugs and vaccines. This prompted another meeting where Rossi presented his findings to the leaders of Flagship Ventures (now Flagship Pioneering) in Cambridge including Flagship founder Noubar Afeyan. Within several months, Rossi, Springer, Langer, Chien and Afeyan formed a new venture they named Moderna. The company name was inspired by combining “MODified” and “RNA”.
We will explore the following in this profile after covering the relevant background:
Vaccines are the beachhead for Moderna to develop transformative medicines.
Moderna provides platform technology to commercialize “apps” for therapies.
Leveraging Kendall Square as ground zero for “patient capital” and biotech innovation.
Messenger RNA technology is not unique to Moderna. The core enabling technology to increase mRNA stability while at the same time decreasing inflammation was discovered at the University of Pennsylvania by Drew Weissman MD PhD and Katalin Kariko PhD. Other known companies pursuing vaccines and drugs with this technology include BioNTech, Translate Bio, CureVac, GritStone Oncology, Kernel Biologics and Arcturus Therapeutics. Moderna and BioNTech are leading this group with commercialized COVID-19 vaccines and robust vaccine and drug development pipelines. COVID-19 variant research and development also provide Moderna and BioNTech advantages with accelerated approvals for “booster” strategies. Moderna and BioNTech may also have first mover advantages based on their scale and safety data to combine seasonal flu and coronavirus vaccines on an annual basis in the future.
But Moderna’s primary challenge is not the competitive threat from other companies, but its own ability to leverage the beachhead it created for synthetic mRNA vaccines to combat infectious diseases into novel therapies for other diseases like cancer. If Moderna can successfully realize its original vision for synthetic biology, the medical and commercial opportunities are substantial.
Background
After founding Moderna in 2010, Noubar Afeyan approached Stephane Bancel regarding the commercial potential for the startup biotech venture. Bancel is a life science leader, chemical engineer and Harvard MBA who at just 34 years old became the CEO of French diagnostics firm BioMerieux. Bancel joined Moderna as founding CEO in 2011. Co-founders Afeyan served as Chairman and Robert Langer served on the Board of Directors.
Moderna raised $40 million from venture capitalists in 2012 while still being years away from testing its science in humans. Bancel also raised $240 million from AstraZeneca for the rights to mRNA that did not exist. Moderna raised more than $1 billion in investments and partnership funds over the next five years and raised more than $2 billion before going public in 2018 in a record-setting and hyped IPO that raised another $600 million at a valuation of about $7.5 billion.
Early research and development efforts at Moderna focused more on developing novel mRNA drugs that could be safely dosed over and over, but animal studies showed the ideal doses were triggering dangerous immune reactions. Lower doses to reduce the immune reactions proved too weak to show any benefits. Moderna pivoted to vaccines that would require only one or two doses to show an effect. Zika, Cytomegalovirus (CMV), Influenza H7N9, HIV, Nipah, Epstein-Barr virus (EBV), Flu and Respiratory Syncytial virus (RSV) are just some examples of vaccine targets in various stages of development at Moderna today.
Vaccines were mostly considered a loss leader for the pharmaceutical industry prior to COVID-19 based on dated technology and long lead times to establish safety data for approval. Pharmaceutical companies were divesting or reducing investments in vaccines to pursue higher margin drug programs. But vaccines offered Moderna a beachhead strategy to establish safety and efficacy data using a repeatable platform technology based on synthetic mRNA. With this technology, the building blocks for any vaccine are just the sequencing of four unique nucleotides and the “printing” process to build a single strand synthetic mRNA molecule. The changes for different vaccine candidates are the sequence of nucleotides and size of the resulting molecules. The end result is mRNA used by the human body to produce a specific protein. For a vaccine, this protein forms an antigen to trigger a specific immune response.
Moderna had already developed the digital and biotechnology infrastructure to design and develop synthetic mRNA molecules for vaccines when the SARS-CoV-2 virus was discovered in Wuhan, China. The SARS-CoV-2 virus was sequenced and published online on January 10, 2020. Moderna’s team was able to move quickly in collaboration with other scientists at the National Institute of Allergy and Infectious Diseases (NIAID) Vaccine Research Center and the University of Texas at Austin’s McLellan Lab to model the sequenced virus and target a novel spike protein to mimic a SARS-CoV-2 infection without exposure to the actual virus. Moderna finalized the synthetic mRNA sequence for what it called mRNA-1273 on January 13 to initiate production and pre-clinical research. Moderna submitted the Investigational New Drug (IND) Application to the FDA for mRNA-1273 on Feb 20, 2020. The Phase 1 dose-escalation clinical trial started on March 16, 2020 in collaboration with the NIAID Vaccine Research Center and the mRNA-1273 Study Group. Non-human primate trials were conducted in parallel with human trials.
The Phase 2 clinical trial started enrollment on May 29 with 300 subjects age 18-55 and 300 subjects age 55-70. The trial was fully enrolled on July 8, 2020. The Phase 3 COVE trial fully enrolled 30,420 volunteers at 99 centers across the United States on October 22, 2020. Two intramuscular injections (50 mL each) of mRNA-1273 or placebo in a 1:1 ratio were administered 28 days apart. The primary end point was prevention of Covid-19 illness with onset at least 14 days after the second injection in participants who had not previously been infected with SARS-CoV-2. The mRNA-1273 vaccine showed a 94.1% efficacy at preventing Covid-19 illness, including severe disease. No safety concerns were identified.
The FDA approved an Emergency Use Authorization for mRNA-1273 on December 18, 2020 and the Moderna COVID-19 Vaccine was soon shipping to medical centers to start vaccinating healthcare workers.
Moderna
Company: Moderna, Inc.
Founded: September 2010, Cambridge, Massachusetts
Founders: Derrick Rossi, Timothy A. Springer, Robert S. Langer, Kenneth R. Chien, Noubar Afeyan
CEO: Stephane Bancel
Nasdaq: MRNA
2020 Revenue: $803 million (+1234.3% Year-over-Year)
Moderna’s mission statement is “Deliver on the promise of mRNA science to create a new generation of transformative medicines for patients.” Moderna believes mRNA is the “software of life”.
Product: Moderna has one commercial product, Moderna COVID-19 Vaccine; the 1st 100 million doses for use in the United States were delivered in March 2021 with emergency use authorization (EUA); authorization for emergency use has also been received by Canada, Israel, European Union, United Kingdom, Switzerland, Singapore and Qatar. Moderna is scaling production to deliver 800 million to 1 billion doses of the vaccine by the end of 2021 and 3 billion doses by the end of 2022.
Strategy: Leverage the fundamental role mRNA plays in protein synthesis to develop a new category of medicines. Digitization is a core attribute and key enabler for Moderna’s business strategy. Moderna will lead clinical programs alone or in collaboration with government agencies like the NIAID and BARDA for Prophylactic Vaccines. Moderna is also open to partner with pharmaceutical companies in areas like cancer and cardiovascular disease to leverage external expertise for specific drug targets and clinical programs.
Moderna has developed an mRNA Design Studio to digitally design mRNA and order mRNA constructs for research. This capability offers Moderna and its partners the opportunity to pursue multiple drug targets in parallel for research and leverage the collective insights to iterate fast and cost effectively. The manufacturing process is the same. The recipe just changes for different drug candidates.
Moderna has also developed an mRNA Early Development Engine internal and external services for GLP toxicology studies, cGMP grade production of drug candidates for human feasibility trials and the design of definitive clinical trials towards FDA submission. These services are available for both internally and externally funded programs.
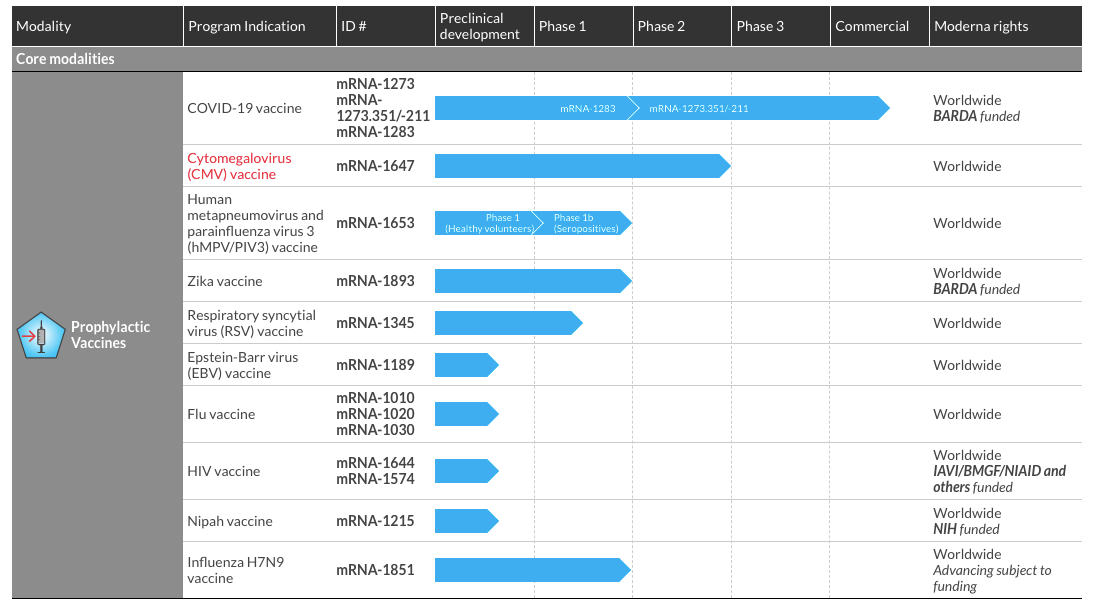
Innovation: Moderna’s development pipeline represents active clinical programs in development to create mRNA medicines for a wide range of diseases and conditions. Prophylatic Vaccines currently represent the largest modality with 1 commercial product for COVID-19 based on vaccine candidate mRNA-1273. Moderna is also leading the development of a cytomegalovirus (CMV) vaccine candidate mRNA-1647 currently active in a Phase 2 clinical trial. mRNA-1647 combines 6 mRNAs in a single vial to encode 2 antigens on the surface of CMV. Four other vaccine candidates for other viruses are in Phase 1 clinical trials and seven other vaccine candidates are in preclinical development for other Prophylatic Vaccines.
Another core modality for Moderna’s development pipeline is Systemic Secreted & Cell Surface Therapeutics. One clinical program in this modality funded by DARPA is therapeutic candidate mRNA-1944 encoding the functional antibody CHKV-24. Moderna has completed a Phase 1 clinical trial in this clinical program for the mosquito-born Chikungunya virus. This Phase 1 trial showed for the 1st time the ability to generate therapeutic levels of a complex protein in humans through the systemic administration of synthetic mRNA to essentially instruct the body to make its own medicines. Two other therapeutic candidates are in preclinical development for autoimmune disorders and one therapeutic candidate for heart failure.
Exploratory modalities include cancer vaccines, immuno-oncology, localized regenerative therapeutics and systemic intracellular therapeutics. Moderna is pursuing a personalized cancer vaccine with candidate mRNA-4157 in a 50/50 global profit sharing partnership with Merck. Moderna personalizes the vaccine by taking a pair of genetic profiles for an individual human subject. One profile is from a biopsy of the tumor and the other is from a vial of healthy blood cells. Moderna algorithms compare the DNA sequences of the two samples and produce a list of 34 targets, each encoding a different mutant protein expressed by the cancer that is predicted to be useful in training the immune system to attack the disease.
Another Moderna innovation encapsulates synthetic mRNA within Moderna’s proprietary lipid nanoparticle (LNP) to deliver the vaccine or therapeutic into cells and protect it from enzymes that would otherwise breakdown the mRNA to limit the desired result.
1. Vaccines are the beachhead for Moderna to develop transformative medicines.
In a prior update [U15], we shared how a number of companies using disruptive innovation like Amazon, Palantir, Square and Tesla used a beachhead market to establish a sustainable business as new ventures. Moderna initially pursued a full slate of approximately 24 drug and vaccine candidates with its mRNA technology to realize its shared vision for synthetic biology and the future of medicine. But challenges with side effects at higher doses and adequate therapeutic efficacy at lower doses led to a pivot to focus on prophylactic vaccines that only require 1 or 2 doses to prevent infectious diseases. This reduced the scope of requirements for clinical success. External funding was also possible with government agencies like BARDA and DARPA. The current development pipeline for prophylactic vaccines is shown in Fig. E8-2.
Vaccine targets become an antigen produced by synthetic mRNA to stimulate antibodies to protect a vaccinated individual from infection by the target virus. Moderna developed a pipeline of prophylactic vaccines and the digital tools to innovate in this domain with its mRNA Design Studio and Early Development Engine. Investments were also made into laboratories to produce clinical-grade vaccine candidates for clinical trials adequate for an FDA submission and early commercial production of an approved vaccine.
When a new respiratory coronavirus emerged in Wuhan, China, Moderna already had a vaccine development engine operating in Cambridge, Massachusetts and an established network of external collaborators. Moderna finalized the design for its mRNA-1273 vaccine candidate for SARS-CoV-2 virus within a few days after its genome was sequenced and published online in China. Moderna was already working with the NIAID at the NIH on coronavirus vaccines so the NIAID was familiar with the Moderna technology and platform. Even though more established pharmaceutical companies had legacy vaccine programs, the US government pursued Moderna’s technology for COVID-19. The US government knew an mRNA vaccine was the fastest commercial path to market to combat a worldwide runaway pandemic. BioNTech was also developing a vaccine for COVID-19 using synthetic mRNA technology.
Unlike BioNTech who partnered with Pfizer to commercialize their COVID-19 vaccine, Moderna was able to accelerate mRNA-1273 clinical trials and scale commercial production internally and with third parties using the capital markets. Moderna will deliver 800 million to 1 billion doses of the COVID-19 vaccine this year and 3 billion doses by the end of 2022. Vaccines will also provide a commercial opportunity for Moderna to add “boosters” against SARS-CoV-2 variants as they emerge and warrant additional protection. The annual flu vaccine could also evolve to include vaccines for both the flu and COVID-19. Moderna is developing a flu vaccine based on mRNA technology. Moderna would have a competitive advantage if they have key technology to prevent illness from the flu and COVID-19.
A successful annual Flu/COVID-19 vaccine program provides an opportunity for Moderna to create a business group or division that is focused only on prophylactic vaccines based on the same mRNA technology shared across all modalities. That business group or division just formalizes the beachhead created when Moderna pivoted to vaccines. Prophylatic vaccines enable Moderna to pursue developing safe and commercially viable transformative medicines.
2. Moderna provides platform technology to commercialize “apps” for therapies.
Therapeutics with synthetic mRNA are also possible when a gene in our DNA does not encode the correct sequence of amino acids to form the correct protein or molecule for a specific function. The lack of function marks the disease. But if a patient’s own body can produce the desired protein using synthetic mRNA, the desired function of the protein can be realized as treatment. Digital technology allows scientists to design and iterate synthetic mRNA as the “code” to produce target proteins for specific clinical programs. Multiple designs can be submitted for synthesis to evaluate different designs and different doses with experiments. The process is scalable to support multiple clinical programs in parallel.
This digital design and streamlined production using the same lines for every vaccine or drug candidate lowers the burden for pre-clinical research and development to down select therapeutic candidates. The scientific throughput is only limited by how many combinations and experiments are required for conclusive outcomes. Alternative biotechnologies can scale commercial production, but are less practical to run 25, 50 and 100-arm experiments to rule out options that are not viable early in pre-clinical research and development.
Moderna was designed to take many shots on goal early in this pre-clinical phase to fail fast starting with molecular designs on a computer and the cloud using machine learning before moving into lab experiments. Only therapeutic candidates that pass this gauntlet progress to the clinic for Phase 1 trials in humans. This has to be attractive to big Pharma as well where the cost to bring new drugs to market continues to increase. Moderna already has active clinical programs with AstraZeneca, Merck and Vertex. But the COVID-19 Vaccine totally changes the game with $19 billion in revenue a year and scaled production to meet the global demand with the capacity to produce 3 billion doses next year. That infrastructure that produces the 1st commercial product can be repurposed towards every other vaccine and drug that follows. They use the same production lines and ingredients, just different code to produce the designed sequence.
One example of a Moderna rate disease clinical program is methylmalonic acidemia (MMA), a rare, life-threatening, inherited metabolic disorder that is most commonly (60% of cases) caused by a deficiency in methymalonyl-CoA mutase (MUT). Newborns missing this protein fail to break down amino acids that then accumulate in the blood and damage vital organs. Standard care includes dietary and palliative care. The only current treatments includes liver or combined liver and kidney transplants. Moderna’s therapeutic candidate mRNA-3704 for MMA that encodes MUT has been granted Fast Track, Orphan Drug, and Rare Pediatric Disease designation by the U.S. Food and Drug Administration (FDA), and Orphan Designation by the European Medicines Agency (EMA).
Moderna enrolled the first human subject with MMA into a Phase 1/2 trial on September 10, 2020 to investigate mRNA-3704 for a deficiency in MUT. This is Moderna’s first rare disease program to begin clinical trial enrollment. Moderna owns worldwide commercial rights for mRNA-3704. Pre-clinical animal studies in mice did not show an increase in markers for liver toxicity or inflammation with repeat dosing. The Phase 1/2 open-label study is designed to evaluate the safety and tolerability of up to 4 increasing dose levels of mRNA-3704 administered via intravenous infusion in patients one year and older with isolated methylmalonic acidemia due to methylmalonyl-CoA mutase (hMUT) deficiency with elevated plasma methylmalonic acid, a key metabolite that accumulates in the disorder.
This is just one example out of thousands of potential rare diseases that have extremely expensive costs to manage patient care without effective treatment. But the risk is even with the success of the Moderna COVID-19 Vaccine, therapeutics could remain elusive due to significant side effects at higher doses and ineffective therapy at lower doses. Moderna will likely need multiple therapeutic candidates pass clinical trials for commercial release. But if Moderna can validate safety and efficacy to reach commercial markets for a growing number of drugs and vaccine, they will continue to reduce unit production costs following Wright’s Law as discussed in [E3.1]. Moderna will keep many clinical programs in-house, but scale commercial operations through partnerships and joint ventures to leverage clinical programs outside the company for specific diseases or categories. Close proxies would be Intel scaling up x86 chip production and sales through its Intel Inside program with many of the market leading computer OEMs or Apple scaling up iPhone production and sales through its App Store and third-party developer program. The success of the latter has led to many “killer apps” reaching the market through Apple’s technology ecosystem.
3. Leveraging Kendall Square as ground zero for “patient capital” and biotech innovation.
Kendall Square is a thriving innovation hub in Cambridge, MA for the pharmaceutical, biotechnology and broader life science industries. The immediate nearby-area includes the Massachusetts Institute of Technology (MIT), Novartis Institutes for BioMedical Research (NIBR), and Broad Institute in Cambridge with Harvard Medical School just across the Charles River in Boston. Boston University, Boston College, Tufts University, many other universities and colleges and many teaching hospitals in the area also contribute to this Kendall Square ecosystem.
Flagship Pioneering (formerly known as Flagship Ventures) was founded within that ecosystem in 2000 as a venture capital firm by Noubar Afeyan and Edwin Kania. Flagship has raised a reported $5.4 billion over 9 funds through June 14, 2021. Flagship is different than most VCs in that it has a history of primarily building innovative biotech companies in-house in addition to making investments in external ventures. Partners typically identify an area of emerging research that is seen as promising for startup innovation. Then they assemble a team around that concept. And then invest in the winners.
Flagship is also an example of “patient capital” that searches diligently for winning ideas to invest very early for maximum returns often as the only outside investor until significant growth capital is needed to scale commercially towards a desired exit. Other VC firms and investors in the area play a less active role in the formation of biotechnology and life science ventures, but can provide critical external pre-seed and seed capital ahead of growth capital. Corporate venture groups also play a critical role to advance key technologies through capital and access to corporate thought leaders.
Kendall Square improves the quality and quantity of these winning ideas in with proximity to Harvard, MIT and other institutional and corporate labs, scientists, professors, business leaders and students. For example, Harvard offers a joint MBA/MS degree program between the Harvard Business School and School of Engineering and Advanced Science. This program recruits rising young leaders in the biotechnology and life science industry who have a desire to start a venture in this area after 2 years of focused study. Harvard Business School also offers the Blavatnik Fellowship in Life Science Entrepreneurship to allow a small number of HBS students to launch such a venture.
Many leading pharmaceutical, biotechnology and life science companies have corporate and/or research offices in the Cambridge area. These attract and retain the talent to advance key clinical programs or leave to join or create new ventures with the infrastructure that supports these pursuits. Trade associations like Massachusetts Biotechnology Council (Mass Bio) also enhance the ecosystem through collaboration and access to various services. The Cambridge Innovation Center (CIC) is the home for thousands of ventures at One Broadway and other locations nearby. Such close proximity to other entrepreneurs and services in the early stages are vital for success. And the local restaurants and coffee shops in Kendall Square offer complementary infrastructure to meet and to get away from the grind for a few minutes.
Moderna has benefitted from many of these Kendall Square attributes including training their Chairman (MIT PhD) and CEO (Harvard MBA), founding science (Harvard Medical School & MIT), funding (Flagship Pioneering), office space (Flagship Pioneering) and initial talent (Flagship Pioneering). Moderna success with the COVID-19 Vaccine has led to significant growth. This has led to a lot of talent joining the company from area companies in the field. Some of these individuals have had significant vaccine experience with commercial development. This will also allow talent to migrate out of Moderna back into these other companies with the knowledge to build clinical programs around synthetic mRNA through corporate development with Moderna.
Conclusion
Moderna’s mission is to “Deliver on the promise of mRNA science to create a new generation of transformative medicines for patients.” The infrastructure Moderna is building to design mRNA vaccine and drug candidates and produce synthetic mRNA in-house for pre-clinical development enables efficient operations to develop and test new molecules. Synthetic mRNA manufacturing also leverages a common process for all vaccine and drug candidates produced. Each candidate mRNA molecule uses the same ingredients. These ingredients are just arranged in different sequences with different lengths to synthesize a unique vaccine or drug candidate. This expedites synthesizing molecules for research, development and commercial production. A 25-arm experiment is feasible with Moderna’s technology if that is needed to advance the science.
But the promise of breakthrough drugs manufactured by a patient’s own body has been challenging. Moderna’s pursuit to treat a rare disease with Alexion Pharmaceuticals was indefinitely delayed. The drug candidate was not determined safe enough to test in humans. Expectations are high for Moderna with the promise of their vision for medicine through synthetic biology, but at times in the past, progress against their mission has been uncertain. Pivoting to vaccines probably saved the company. When SARS-CoV-2 emerged in China, Moderna was well positioned and ready to act decisively with active clinical programs already pursuing prophylactic vaccines for a number of years.
Moderna also built relationships with scientists working on coronaviruses and the NIAID’s Vaccine Research Center at the NIH for infectious diseases. Moderna’s advanced infrastructure allowed it to design the COVID-19 vaccine candidate mRNA-1273 in collaboration with the NIAID in about 48 hours after the SARS-CoV-2 genome sequence was first published online. The IND for mRNA-1273 was submitted to the FDA only 5 weeks after the vaccine candidate was designed because Moderna already knew the process and clinical trial strategy for a vaccine for an infectious disease from prior work. The Phase 1 clinical trial on mRNA-1273 in human subjects started 8 weeks after the molecule was designed. The Phase 3 clinical trial in over 30,000 human subjects with 2 doses of mRNA-1273 showed 94.1% efficacy at preventing COVID-19 illness, including severe disease. An emergency use authorization for the vaccine was issued by the FDA less than 12 months after the genome for the target virus was first published. Moderna released results for its first profitable quarter on May 6, 2021.
Moderna is not the only biotech company with mRNA vaccines and therapeutics. BioNTech working with Pfizer has its own FDA Emergency Use Authorization for a COVID-19 vaccine. Pfizer has helped scale the BioNTech vaccine faster across more global markets versus Moderna working alone with the capital markets. Pfizer and BioNTech share costs and profits 50/50 for their vaccine. Moderna does not have a pharmaceutical partner for the Moderna COVID-19 Vaccine, but was under contract as of December 2019 with the NIH to develop vaccines for coronaviruses with joint ownership for both parties. Four NIH scientists have also filed a provisional patent on the vaccine. These rights could be used at some point by the US government to license the COVID-19 vaccine IP to other companies if Moderna does not act in the best interest of the US tax payers. But as long as Moderna is investing into building out scaled commercial production plus pre-clinical development into vaccine improvements with SARS-CoV-2 variants, the US government would likely not pursue using its rights to license the vaccine to other manufacturers.
Moderna could generate an estimated $60 billion ($20 billion x 3 years) in revenue over the first 3 years of the COVID-19 Vaccine. That is quite a beachhead market to build and sustain a new biotechnology venture. How Moderna leverages that beachhead for growth will likely determine its fait as either just another biotech with a focus on vaccines or as the Tesla of medicine.
Best,
Stephen
If you are not a subscriber, please consider doing to get updates on this topic and more.
I’m long MRNA mentioned in this story. Nothing in this post is intended to serve as financial advice. Do your own research.